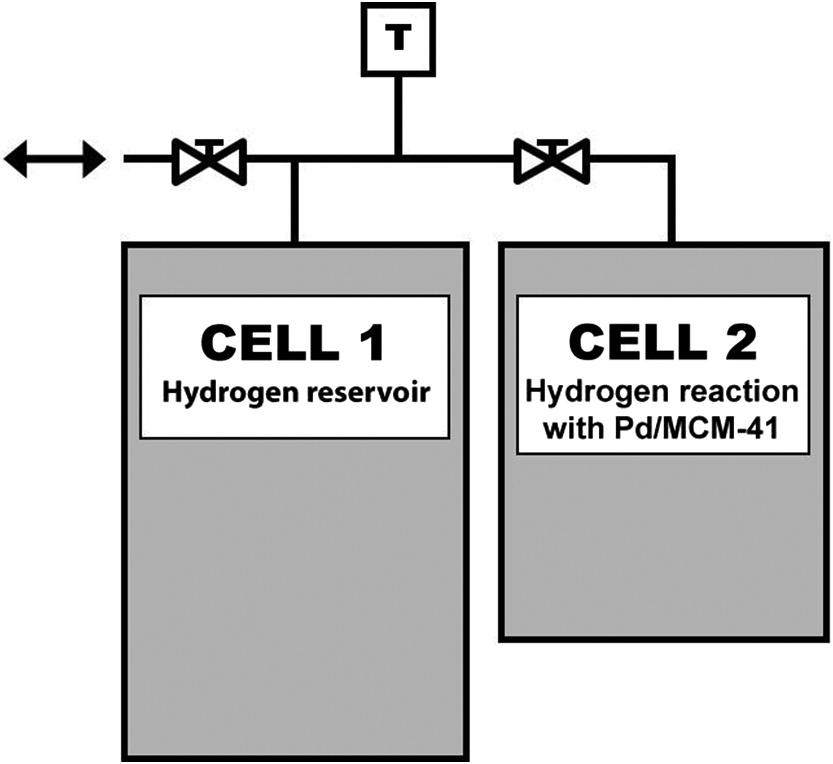
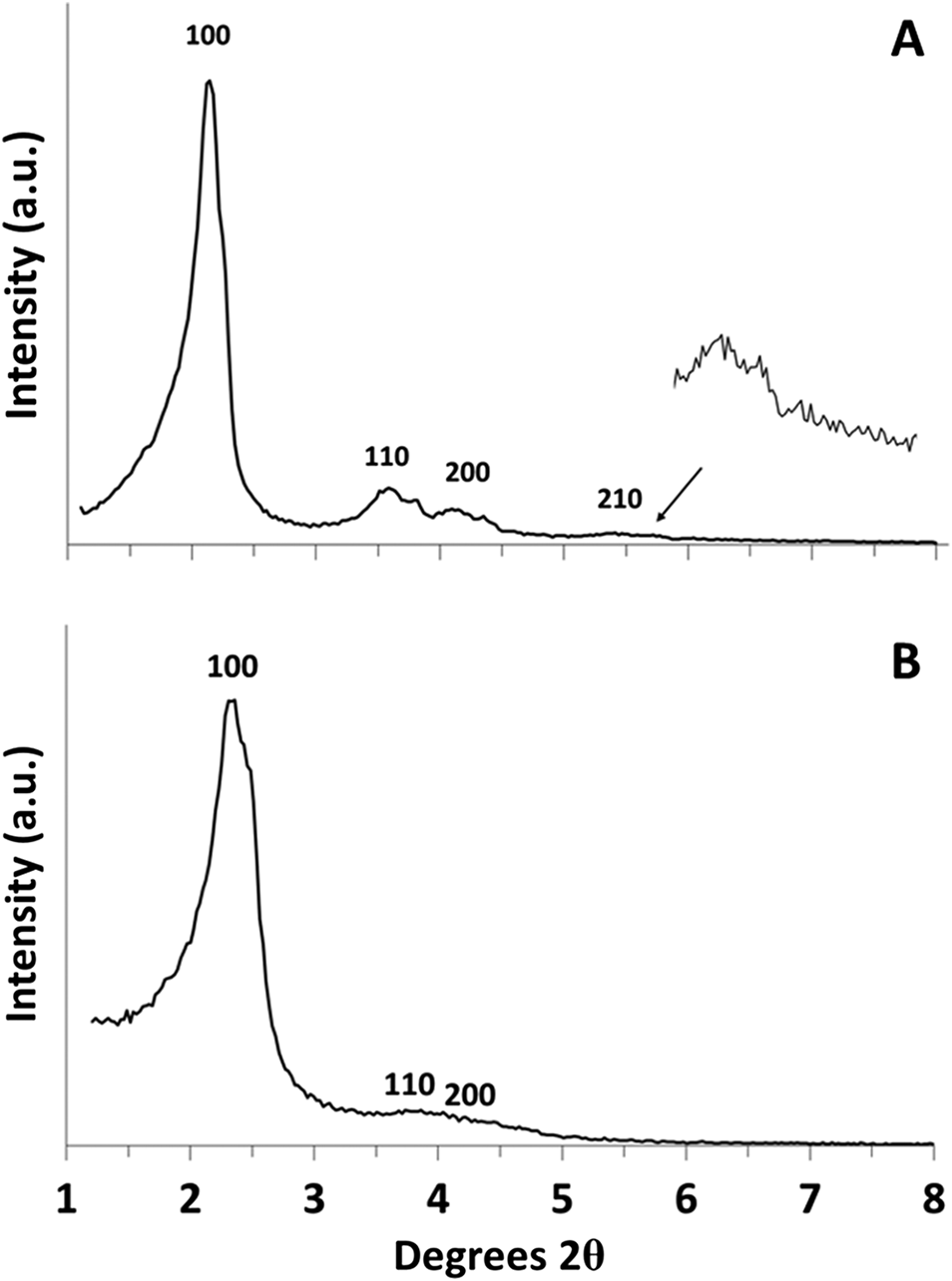
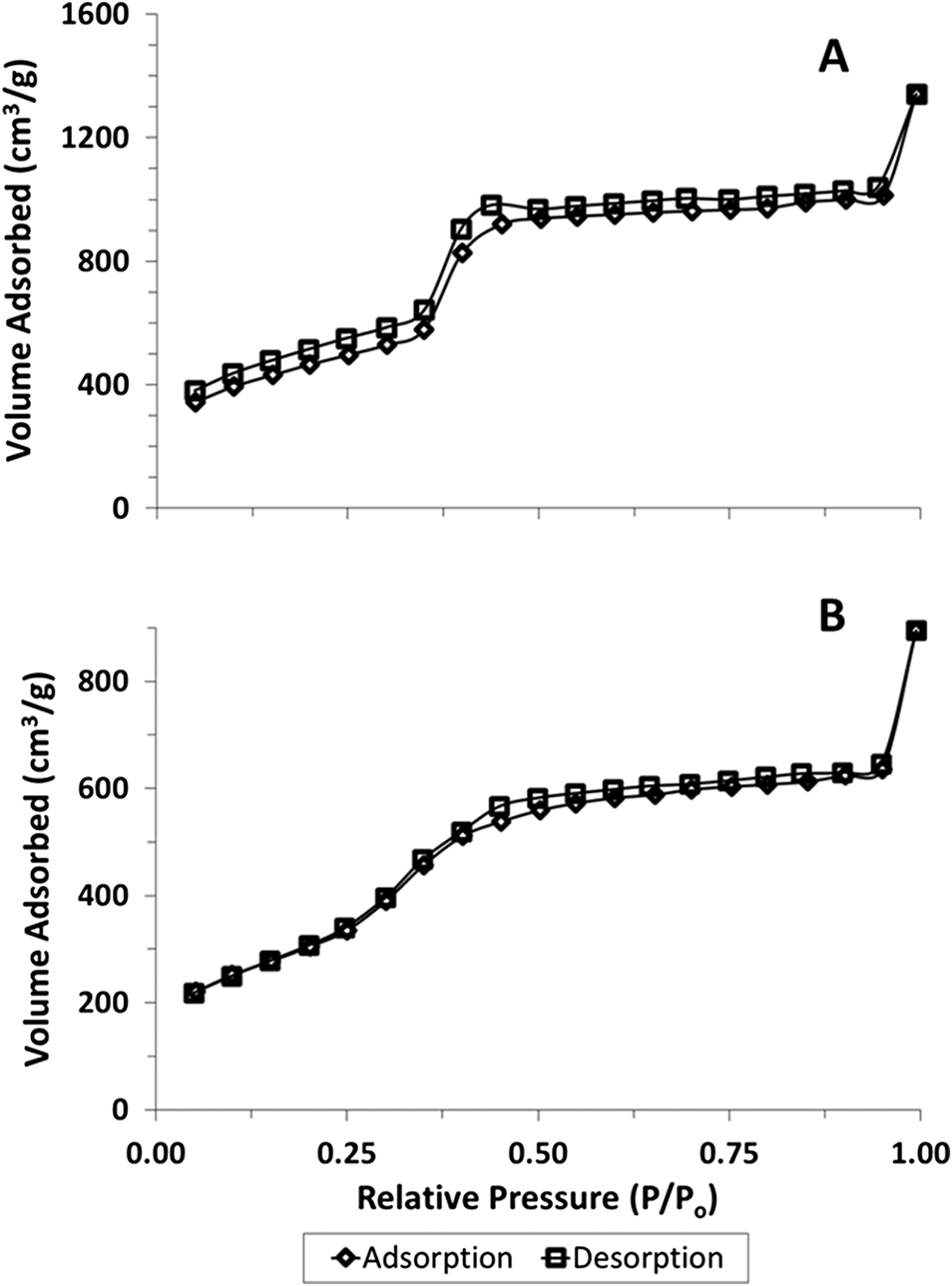
Hydrogen absorption experiments
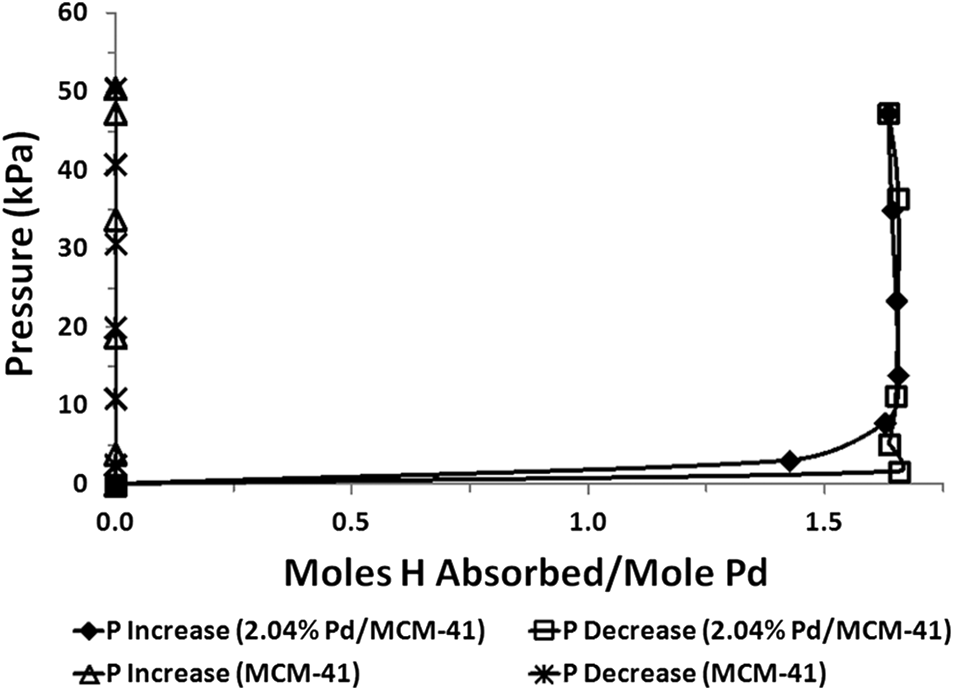
The maximum extent of hydrogen absorption was determined from the steady-state portion of the P–C isotherm, where further increases in hydrogen pressure produced no change in H/Pd. Steady state was defined as points on the curve exhibiting less than 10% change from each previous, lower pressure point. This agreed closely with the earlier identification of steady-state portions of isotherms by visual inspection. Typical results for un-palladized, calcined MCM-41 are shown in
Fig. 7 as a plot of pressure versus moles of absorbed hydrogen. Plotted results confirm no measurable sorption of hydrogen by MCM-41 support material.
A typical P–C isotherm from one experimental run using 2.04% Pd/MCM-41 is also shown in
Fig. 7. Absorption occurred rapidly, with steady-state pressure generally achieved within 10 min of introducing hydrogen to CELL 2 (
Fig. 1). Hysteresis shown by H/Pd results at lower pressures (<0.1 atm) during depressurization stage was typical for all runs and Pd/MCM-41 samples. Consistency of maximum H
2(g) absorption between experimental runs using the same Pd/MCM-41 sample indicated that absorbed H
2(g) was released when the sample was put under vacuum between runs.
The appearance of hysteresis in H/Pd results upon lowering
suggests a two-phase field during absorption/desorption and a phase transition in the metal (
Sachs et al. 2001;
Pundt et al. 2004). Similar hysteresis results of experiments with Pd reported by other researchers were caused by a difference in the formation pressure of the metal hydride phase relative to the decomposition pressure (
Flanagan et al. 1980). This phase transition results from lattice expansion and tensile stress as increasing amounts of hydrogen enter the Pd lattice and metal hydride is formed with corresponding lattice contraction during dehydriding (
Flanagan et al. 1975,
1980;
Flanagan and Oates 1991;
Sachs et al. 2001). Misfit dislocations of the lattice as reported in bulk Pd are not expected in particles with diameter less than 4 nm (
Ichinose 2000;
Sachs et al. 2001). Instead, hysteresis in smaller particles is caused by a coherency, or internal elastic stress in the metal, resulting in different chemical potentials during loading and unloading of hydrogen from Pd, thus forming a hysteresis loop (
Schwarz and Khachaturyan 1995;
Sachs et al. 2001;
Pundt et al. 2004).
Hydrogen pressurization pretreatment of Pd/MCM-41 samples (described above) removes the oxide layer on Pd, leaving a relatively reduced Pd metal. Reduced Pd on Pd/MCM-41 samples prepared following the method of
Koh et al. (1997) should yield an average Pd particle size of 2–2.5 nm with good distribution within MCM-41 pores. It is proposed that hysteresis in this case results from differing chemical potentials during loading and unloading of hydrogen from Pd in samples, rather than the creation of misfit dislocations as occurs in larger diameter Pd particles.
Hydrogen absorption experiments for each Pd/MCM-41 sample were repeated a minimum of five times to demonstrate the degree of reproducibility of P–C isotherms. Results of simplified measurements are also included in this document. In these cases, only one injection of H2(g) was made and the uptake recorded. Maximum uptake of H2(g) measured in this fashion should not differ from that determined via isotherms generated from successive injections of H2(g). Hydrogen absorption results for 0.81% and 2.30% Pd/MCM-41 samples were obtained in this way.
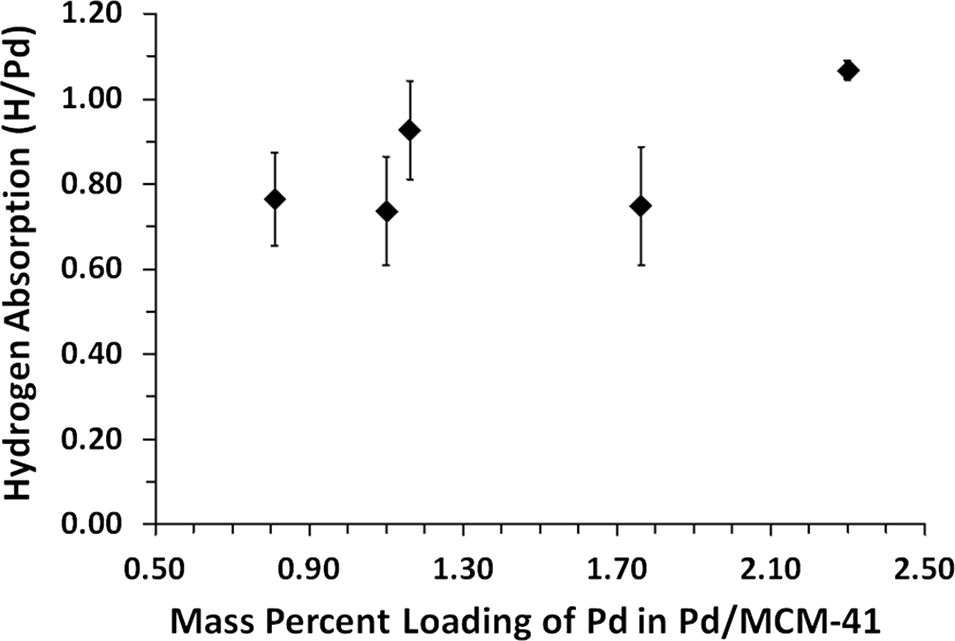
Average H/Pd values of all steady state points on P–C isotherms for each Pd/MCM-41 sample were plotted versus the mass percent loading of Pd (
Fig. 8). Average absorption at all mass percent Pd loadings was 0.85 ± 0.18 H/Pd with an apparent, overall trend of increasing absorption as mass percent of Pd on MCM-41 increased from 0.81% to 2.30%. Maximum average hydrogen absorption for a single mass percent loading of Pd was 1.07 H/Pd for the 2.30% Pd/MCM-41 sample. Minimum average hydrogen absorption for a single mass percent loading of Pd was 0.74 H/Pd for the 1.10% Pd/MCM-41 sample. The
R 2 value of the trendline applied to the H/Pd versus mass percent loading Pd plot was 0.424. The critical
R 2 value for five data points at a 95% confidence level is 0.878. Therefore, the apparent increase in H/Pd as mass percent Pd loading increases was deemed to be non-significant. Results of similar experiments using Pd black showed an absorption of 0.79 ± 0.09 H/Pd, which was 30% less than uptake by 2.30% Pd/MCM-41.
Hydrogen solubility in bulk Pd has been calculated as
S = 27.1
P 0.5 according to Sieverts’ law, where
S is in mol/m
3 and
P is in Pa (
Sachs et al. 2001).
Boudart and Hwang (1975) measured hydrogen absorption of 0.50 H/Pd in small Pd particles supported on SiO
2 and Al
2O
3 at 303 K.
Jewell and Davis (2006) reported 0.03 H/Pd in α-phase Pd and 0.6 H/Pd in β-phase at room temperature.
Pundt et al. (2004) reported that bulk Pd absorbed 0.6 H/Pd at room temperature, whereas Pd surfaces can absorb up to 1 H/Pd.
Kuji et al. (2002) performed experiments with nano-sized and coarser-grained Pd at 298 K. They recorded absorption of just over 0.7 H/Pd at 100 kPa for coarse-grained samples, while nano-sized Pd samples of diameter 8 nm absorbed between 0.45 and 0.50 H/Pd.
Sachs et al. (2001) studied surfactant- and polymer-stabilized nano-clusters of Pd at room temperature. They reported absorption between 0.40 and 0.65 H/Pd at 100 kPa for surfactant-stabilized clusters of diameter 2, 3, and 5 nm. Researchers have noted that small Rh metal clusters less than 1 nm in diameter can absorb up to eight atoms of deuterium per metal atom; however, the maximum absorption reported for Pd was lower at 3.25 mol of deuterium per mole of neutral Pd atoms and approaches unity as Pd cluster size increases (
Cox et al. 1990;
Sachs et al. 2001;
Pundt et al. 2004).
In the only published paper to date on the H/Pd ratios in palladized MCM-41,
Prasanth et al. (2010) prepared 3.7% by weight Pd-incorporated, hydrothermally synthesized MCM-41 and recorded the absorption of 0.306 H/Pd (35.1 cc H
2/g) at 303 K. In their material, Pd
2+ was incorporated into the MCM-41 structure during synthesis rather than being deposited onto the surfaces and within pores. Additionally, their mass percent of Pd in MCM-41 samples was greater than the 2% maximum used in this current study. Both these aspects of
Prasanth et al.’s (2010) absorption study may account for their substantially lower observed H/Pd results.
A combined comparison of all published results is difficult due to the differences in reaction temperatures, support materials, mass of Pd, and preparation techniques. However, average absorption by Pd/MCM-41 at 298.2 ± 0.1 K and Pd mass percent loadings between 0.78% and 2.04% was higher than results published by other researchers using different Pd preparations and support materials. The enhanced deuterium absorption by metal particles as their diameter decreased below 1 nm reported by
Cox et al. (1990) implies the possibility of similar enhancements for other small-sized transition metals. This suggests that further enhancements in hydrogen uptake may be achieved using exceptionally small particles of Pd. Enhanced absorption by Pd/MCM-41 compared to other previously published results using other Pd materials may be explained by either a larger proportion of subsurface sites in nano-sized Pd particles in Pd/MCM-41 relative to surface and bulk sorption sites or by a spillover of H
2(g) onto the surfaces of the MCM-41 support material (
Eriksson and Petersson 1994).