Altered acrylic acid concentrations in hard and soft corals exposed to deteriorating water conditions
Abstract
Introduction
Materials and methods
Animal management, water quality monitoring, and biopsy sampling
| Water quality parameter | Mean | Range | Mode | % of total readings |
|---|---|---|---|---|
| Temperature (°C) | 25 | 24–27 | 24 | 50 |
| Specific gravity | 1.024 | 1.023–1.025 | 1.024 | 50 |
| pH | 8.2 | 8.1–8.4 | 8.2 | 67 |
| Ammonia (mg/L) | 0 | 0–0 | 0 | 100 |
| Nitrite (mg/L) | 0 | 0–0 | 0 | 100 |
| Nitrate (mg/L) | 0 | 0–0 | 0 | 100 |
| Calcium (mg/L) | 430 | 400–500 | 400 | 50 |
| Phosphate (mg/L) | 0 | 0–0 | 0 | 100 |
Note: All water quality test kits used were in date and stored properly. Water quality testing was routinely completed by a small number of individuals.
Sample processing and NMR data acquisition
Data processing
Studies of coral tissue dissociated from zooxanthellae
Results
| Metabolites | Reference chemical shift (ppm) and peak shape (br s, d, dd, ddd, dq, m, q, or s) |
|---|---|
| Alcohols | |
| Methanol | 3.34 (s) |
| Alkaloids | |
| Trigonelline | 4.428 (s), 8.072 (m), 8.826 (m), 9.114 (s) |
| Amino acids | |
| Alanine | 1.47 (d), 3.76 (q) |
| Betaine | 3.25 (s), 3.89 (s) |
| Carnitine | 2.43 (dd), 3.21 (s), 3.42 (m), 4.56 (br s) |
| Glycine | 3.54 (s) |
| Valine | 0.976 (d), 1.029 (d), 2.261 (m), 3.601 (d) |
| Amino acid derivatives | |
| N,N-Dimethylglycine | 2.91 (s), 3.71 (s) |
| Alkylamines | |
| Dimethylamine | 2.7 (s) |
| Trimethylamine | 2.89 (s) |
| Fatty acids | |
| Acetate | 1.91 (s) |
| Acrylic acida | 5.89 (dd), 6.14 (q), 6.40 (dd), 12.08 (s) |
| Isovalerate | 0.9 (d), 1.94 (dq), 2.05 (d) |
| Guanidines | |
| Methylguanidineb | 2.833 (s), 3.366 (s) |
| Organic acids | |
| Formatec | 8.44 (s) |
| Lactate | 1.32 (d), 4.1 (q) |
| Malonate | 3.11 (s) |
| Purine bases | |
| Adenine | 8.11 (s), 8.12 (s) |
| Pyrimidine bases | |
| Thymine | 1.86 (s), 7.37 (s) |
| Quaternary ammonium | |
| Choline | 3.189 (s), 3.507 (dd), 4.056 (ddd) |
| Sulfonyls | |
| Dimethyl sulfoneb | 3.138 (s) |
Note: Metabolites were identified using Chenomx NMR Suite 7.63 (Chenomx, Edmonton, Alberta, Canada), the Human Metabolome Database (Wishart et al. 2013), the Yeast Metabolome Database (Jewison et al. 2012), the Spectral Database for Organic Compounds (National Institute of Advanced Industrial Science and Technology 2016), and reference chemical shifts predicted from ACD Labs 12.0 1D NMR Processor (Advanced Chemistry Development, Toronto, Ontario, Canada). br s, broad singlet; d, doublet; dd, doublet of doublets; ddd, doublet of doublet of doublets; dq, doublet of quartets; m, multiplet; q, quartet; s, singlet.
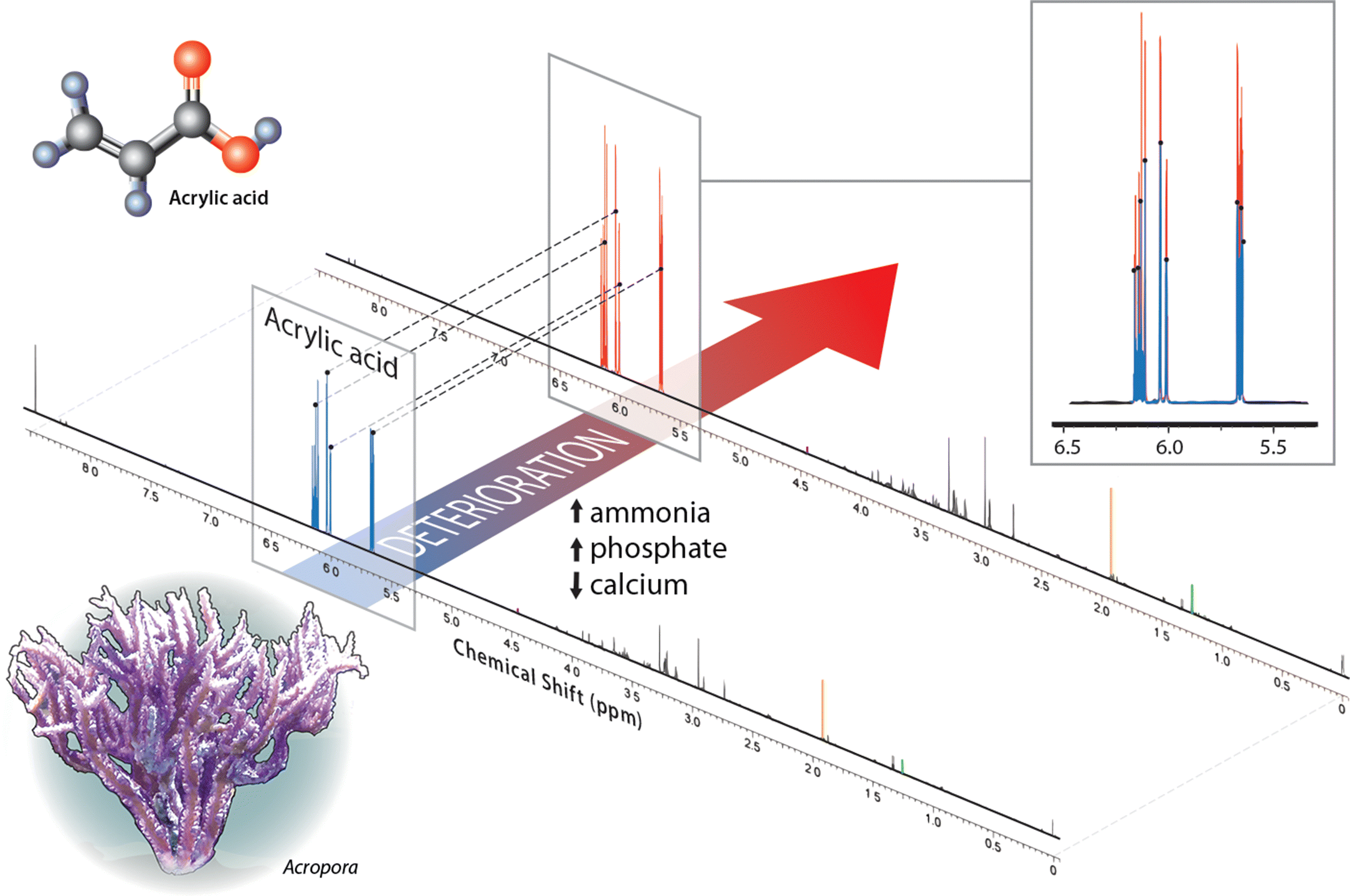
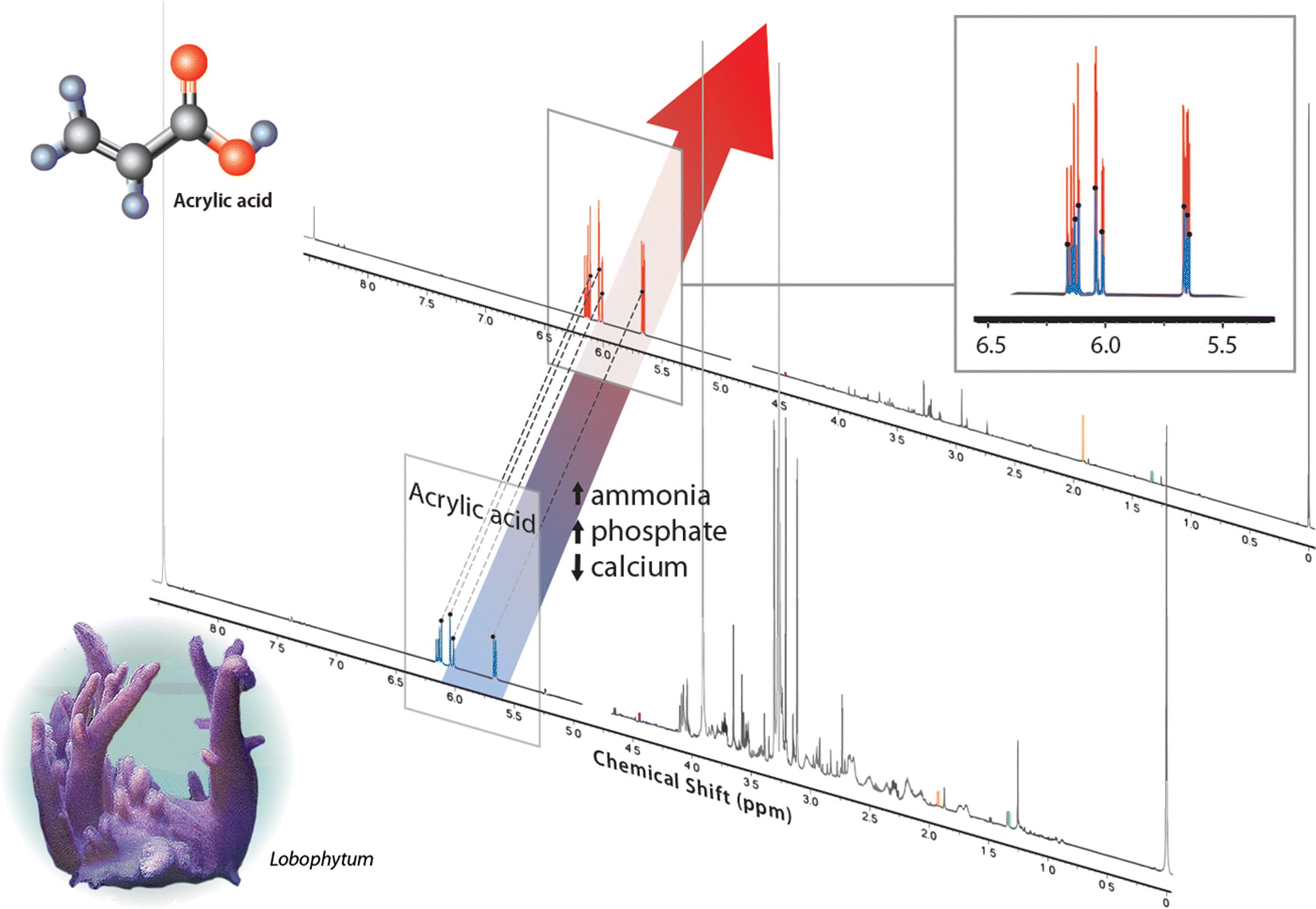
| Percent change (deteriorated/good) | Good water (area under curve) | Deteriorated water (area under curve) | |
|---|---|---|---|
| Acropora sp. | |||
| Acrylic acid | 180 | 0.2133 | 0.3287 |
| Acetate | 180 | 0.0108 | 0.0164 |
| Lactate | 0 | 0.0042 | 0.0033 |
| Thymine | −10 | 0.0008 | 0.0006 |
| Trigonelline | 250 | 0.0004 | 0.0009 |
| Sample weight (g) | — | 0.430 | 0.363 |
| Lobophytum sp. | |||
| Acrylic acid | 190 | 0.0532 | 0.2134 |
| Acetate | 180 | 0.0029 | 0.0108 |
| Lactate | −70 | 0.0059 | 0.0042 |
| Thymine | −90 | 0.0043 | 0.0008 |
| Trigonelline | 0 | 0.0002 | 0.0004 |
| Sample weight (g) | — | 0.220 | 0.454 |
Note: The sum of areas under the curve for assigned 1H-NMR spectral peaks after normalization of the spectrum and adjustment based on the TSP internal standard for each compound of interest are presented. Percent changes are calculated for each compound for a given coral species by dividing the area under the curve for deteriorated water coral samples by the area under the curve for coral samples from good water and then multiplying by 100 times the ratio of the weights of the good over the deteriorated water coral samples originally processed for spectroscopy.
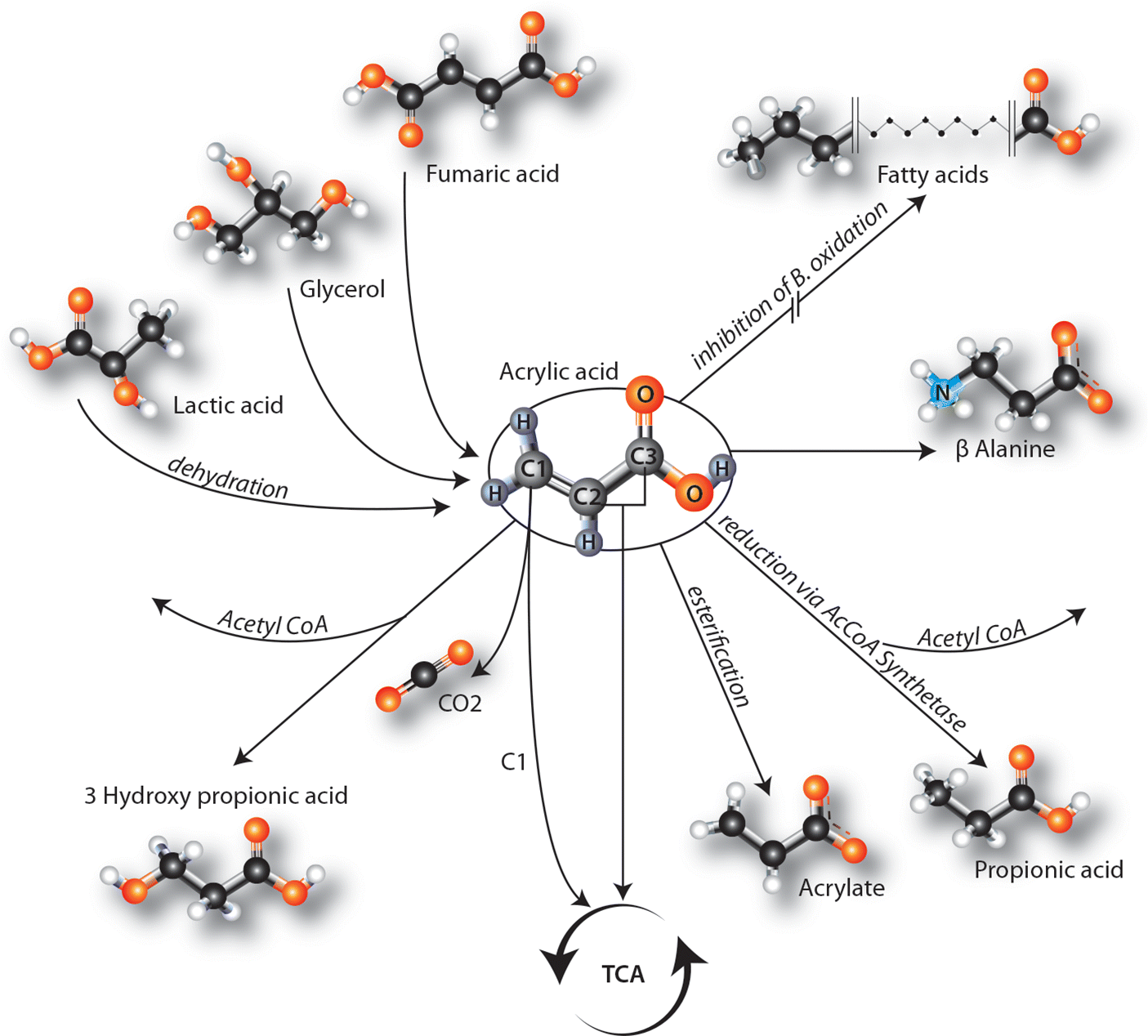
Discussion
List of abbreviations
- NMR
- nuclear magnetic resonance spectroscopy
- 1H-NMR
- proton nuclear magnetic resonance spectroscopy
- 13C-NMR
- carbon nuclear magnetic resonance spectroscopy
- Acetyl-CoA
- acetyl coenzyme-A
- DMS
- dimethylsulfide
- DMSP
- dimethylsulfoniopropionate
- TCA
- tricarboxylic acid
- TSP
- trimethylsilylpropanoic acid
Acknowledgements
References
Information & Authors
Information
Published In

History
Copyright
Data Availability Statement
Key Words
Sections
Subjects
Plain Language Summary
Authors
Author Contributions
Competing Interests
Metrics & Citations
Metrics
Other Metrics
Citations
Cite As
Export Citations
If you have the appropriate software installed, you can download article citation data to the citation manager of your choice. Simply select your manager software from the list below and click Download.
