Introduction
Nature’s variability has been shown to play a major role in the stability of food webs and whole ecosystems (
Levin 1998;
McCann and Rooney 2009;
Bernhardt et al. 2020). Within food webs, spatial and temporal variability contributes to food web stability by providing multiple energy channels and diverse resource pools that mobile consumers can forage across (i.e., consumptive portfolio effect;
McCann and Rooney 2009;
McMeans et al. 2015;
Gutgesell et al. 2022). Generalists that couple multiple habitats and omnivores that couple multiple trophic levels can capitalize on fluctuating resource densities if they can rapidly switch their foraging to the most abundant resource, thereby accessing a consistent aggregate resource supply. Such switching produces shifting interaction strengths that release predation pressure on lower density resources, helping weaken energy flows and contributing to overall food web stability (
McCann and Rooney 2009;
McMeans et al. 2015;
Gutgesell et al. 2022). In contrast, anything that homogenizes resources in space and time or inhibits a consumer’s ability to rapidly shift with changing resource densities potentially threatens the stability of whole food webs.
Human induced stressors, such as land-use change, agriculture, and climate change have the potential to alter spatial and temporal variability in ways that erode stabilizing mechanisms that maintain ecosystem structure and function (
Gutgesell et al. 2024). Such alterations reshape how energy flows through food webs by modifying interaction strengths, generating or removing interactions (i.e., altering food web topology), or shifting biomass structures, thus fundamentally “rewiring” energy flow through food webs (
Blanchard 2015;
Bartley et al. 2019). Theory has shown that rewiring that generates shifts toward single-energy channel food webs and increasing interaction strengths and/or energy flow can reduce the stability and function of whole food webs and ecosystems (
Oksanen et al. 1981;
Rip and McCann 2011;
McCauley et al. 2018;
Bartley et al. 2019). As an example, it is well known from theory that increasing productivity (as may be expected from nutrient run-off in agricultural landscapes,
Allan 2004) increases energy flow through food webs and can lead to instability (i.e., mean and variance driven stability loss;
Rosenzweig 1971;
McCann et al. 2021). Therefore, it is important to understand how different types of anthropogenic changes rewire food webs to gain insight into the resulting implications for food web stability and function.
The food webs of temperate headwater streams are strongly structured and stabilized by temporal (i.e., seasonal) variability in allochthonous and autochthonous resource availability (
Nakano and Murakami 2001;
Takimoto et al. 2002). The seasonal asynchronous flux of in situ aquatic resources in the spring and early summer and terrestrial subsidies in fall provide a consistent resource supply to generalist fish over time (
Nakano et al. 1999;
Nakano and Murakami 2001;
Takimoto et al. 2002;
Richardson et al. 2010;
Larsen et al. 2016). Generalist fish that respond to these seasonally-fluctuating resource densities to capitalize on the dominant resource couple aquatic and terrestrial energy channels and exhibit temporally shifting interaction strengths (
Nakano and Murakami 2001;
Richardson et al. 2010). The coupling of these fast (aquatic) and slow (terrestrial) energy channels across season by generalist consumers has been shown to be a highly stabilizing structure in stream food webs (
Takimoto et al. 2002). Therefore, alterations in seasonal aquatic and terrestrial resource inputs can rewire food web interactions of consumer fish, ultimately impacting the stability of stream food webs.
Agricultural land cover change has the potential to rewire stream food webs by altering the seasonal asynchronous flux of aquatic and terrestrial resources and shifting systems towards single-energy channel dominated webs. Removing riparian zones have been shown to reduce the amount of terrestrial in-fall (
Kawaguchi and Nakano 2001;
Little and Altermatt 2018;
Grunblatt et al. 2019), and changes such as nutrient loading can increase aquatic productivity (
Finlay 2011;
Griffiths et al. 2013). Combined, these changes can alter relative resource availability across seasons for generalist fish consumers and may shift feeding interactions and biomass structure in ways that may be destabilizing. Previous research has shown that within a single season (summer), variations in riparian buffer size can alter trophic interactions of a generalist fish species, creek chub (
Semotilus atromaculatus), by reducing reliance on the terrestrial energy channel (
Champagne et al. 2022). Therefore, it would be expected that a disruption in the asynchrony of terrestrial and aquatic resource availability could reduce seasonal switching across energy channels, shifting stream food webs towards single-energy channel dominated systems with increased interaction strengths/energy flux (
Effert-Fanta et al. 2022). Homogenized stream habitats with large increases in aquatic productivity may also increase the productivity of fish communities and potentially shift community biomass structure towards high abundances of small, fast growing generalist fish (
Casatti et al. 2012;
Gutgesell et al. 2024). Combined, these changes would rewire the structure of stream food webs through time in ways that may generate increased instability.
Here, we aim to determine the potential for agriculture to rewire the seasonal structure of temperate stream food webs using a combination of stable isotopes, stomach contents, and fish community biomass structure measured across a gradient of agricultural impact in southwestern Ontario, Canada. We hypothesize that agricultural land-use change will rewire seasonal stream food web structure by altering the asynchrony of seasonal aquatic and terrestrial resource availability, ultimately driving shifts towards productive single-energy channel dominated food webs. To address this hypothesis we test the following predictions: (1) agricultural land-use change will reduce terrestrial resource availability in summer and fall through the removal of riparian zones and increase in aquatic vegetation production through nutrient run-off in the spring and summer (
Fig. 1); (2) temporal changes in resource availability will alter fish trophic interactions such that they reduce % terrestrial energy use (% TEU), reduce seasonal switching from aquatic to terrestrial pathways, and increase omnivory (i.e., increase reliance on lower trophic resources, as reflected by lower consumer trophic position (TP) estimates). Collectively these changes in trophic interactions will seasonally alter the trophic niche of generalist fish consumers (
Fig. 1); and (3) the increased reliance on the productive, aquatic energy channel by fish will shift fish community biomass structure towards high abundances of small, fast-growing individuals.
Methods
Study system
The study was conducted in three headwater streams (stream order ≤ 3) along the north shore of Lake Erie, Ontario, a region with high cover of agricultural land (Fig. S1). We collected seasonal stable isotope, stomach contents, fish community composition, and supporting stream characteristic data from three streams spanning a gradient from low to high agricultural impact. Previous work in this study system examined mean food web responses to agricultural impact across 20 streams and demonstrated the importance of local agriculture in driving resultant food web responses (
Champagne et al. 2022). To unpack impact of agricultural land-use change on seasonal food web structure we selected representative sites at low, middle, and high end points of percent local agricultural gradient in (Fig. S2;
Champagne et al. 2022). The low-impact site is representative of protected or conserved streams with intact riparian buffers and relatively low agricultural activity in its watershed. The mid-impact site is representative of agricultural sites with reclamation or restoration of marginal lands (e.g., farms part of the Alternative Land-use Program; ALUS Canada,
www.alus.ca), with more intact riparian zone, yet high agricultural activity in the watershed. The high-impact site is representative of more intensive conventional agricultural practices where forested riparian zones have been removed and agricultural activity occurs directly next to the stream.
Land use
Land-use composition was characterized at three spatial scales (local, riparian, and watershed) at each stream to characterize agricultural impacts and estimate relative aquatic and terrestrial resource availability (Fig. S3). Local % agriculture and local % forest cover were calculated in a 250 m radius around each fish sampling reach from the 2015 Agriculture and Agri-Food Canada (AAFC) semi-decadal land-use data using the sf and raster packages in R (
Pebesma 2018;
Hijmans 2025). Agricultural land comprised of cropland, annual cropland, and managed grasslands, and forest land was comprised of forest, forest wetland, forest regenerating after harvest < 20 years, forest wetland regenerating after harvest < 20 years, and forest regenerating after fire < 20 years (
AAFC 2015).
Riparian land-use composition was calculated as the percent agricultural and percent forest land cover within a 100 m buffer surrounding the full river network upstream of each fish sampling reach. ArcGIS v. 10.6.1 was used to generate shapefiles of the 100 m buffer zone upstream of the sampling reach and % agriculture and % forest cover within the riparian buffer zone were calculated using the 2016 Ontario Land Cover Compilation v.2.0 data (
OLCC 2016). Additionally, average riparian buffer width (the width of canopy dominated vegetation along the streambank) was measured using aerial photography from Google Earth Pro (Google Earth Pro, Google Inc., Mountain View, CA, 155 USA). The width of forested canopy perpendicular to the left and right banks were measured at 0 m (i.e., at sampling reach), 0.5 km, 1 km and 2 km upstream of the sampling reach. Measurements were averaged to obtain an average riparian buffer width for each stream.
Watershed land-use composition was calculated as the % agricultural and % forest land within each streams’ individual watershed. Watershed shapefiles were obtained from the Ontario Flow Assessment Tool (OFAT; OMAFRA,
www.ontario.ca/page/watershed-flow-assessment-tool) and imported into ArcGIS (v. 10.6.1). The % agriculture and % forest cover within each watershed was calculated using the 2016 Ontario Land Cover Compilation v.2.0 data (
OLCC 2016).
Lastly, as agricultural land-use change has been shown channelize streams, which contributes to within stream habitat homogenization (
Petersen 1992;
Allan et al. 1997), we calculated sinuosity of each stream as an additional indicator of agricultural land-use change. Sinuosity was calculated by dividing the 5 km distance upstream from the sampling reach by the straight-line distance between the two points (
Fitzpatrick et al. 1998), using aerial photography from Google Earth Pro.
Physical stream characteristics
Stream characteristics were measured to help characterize agricultural impacts and examine changes in stream condition. For each stream, wetted width (cm), water depth (cm), and hydraulic head (estimate of relative flow velocity, cm;
Stanfield 2013) were measured twice a week from the beginning of May to the beginning of November 2018 (3 May to 5 November 2018). Each wetted width measurement was taken at the same point in the middle of the established fish sampling reach (see details on sampling reach selection in fish sampling methods below). Each water depth and hydraulic head measurement was recorded from the mid-channel point where wetted width was measured. Hydraulic head (mm) was measured as the difference in height of water between the front and back of a vertically held ruler placed at a right angle to the flow of water and is indicative of water velocity (
Stanfield 2013). Mean and standard deviation for each metric was calculated and is reported in
Table 2.
Total nitrogen (N), total phosphorous (P), and % algal cover was measured at each of the streams monthly from June to August 2018. For N and P analysis, 1 L water samples were collected from the upstream, middle, and downstream points of the sampling reach. All samples were stored at −20 °C until analysis. N was measured as total Kjeldahl nitrogen (TKN, which contains organic nitrogen, ammonia, and ammonium) and was determined using segmented flow colourimetry, with a detection limit of 0.05 mg/L. P concentrations were determined using inductive coupled mass spectrometry with a detection limit of 0.003 mg/L. All nutrient analysis was conducted by SGS Canada (SGS Canada Inc., Lakefield, Ontario, Canada). % algal cover was estimated based off visual estimation of the stream, where coverage was categorized as 0%–20%, 20%–40%, 40%–60%, and 60%–100%. To generate a mean summer % algae cover, the mid-point of each range was used and then an average across the 3 months was calculated.
Fish sampling
Sampling of fish communities was conducted at each stream once in spring, summer, and fall 2018. Spring sampling was conducted from 28 to 30 May, summer sampling was conducted from 30 July to 1 August, and fall sampling was conducted from 23 to 26 October. Fish were collected using a triple-pass electrofishing method based on the Ontario Stream Assessment Protocol Version 9.0 (
Stanfield 2013). At each site a 40 m reach was selected to include the best representation of all available habitats present (S-bend, riffle-pool-riffle sequence, or straight channel). Each reach was blocked and all fish collected were held or released downstream of the blocked sampling reach until all three passes were complete. The same reach in each stream was sampled each season.
All fish caught were identified and weighed to examine fish community composition and biomass structure. For examining changes in food web structure, creek chub were selected as the focal species as these fish are highly generalized feeders and can operate as the top predator in streams when other more typical top predators (e.g., brook trout (
Salvelinus fontinalis) and rock bass (
Ambloplites rupestris)) are absent (
Holm et al. 2009;
Champagne et al. 2022). Creek chub are also silt-tolerant and have been shown to replace brook trout as the dominant fish species in streams with higher land-use alteration in Southern Ontario (
Martin 1984). No trout or bass species were caught at any of the sampling sites (Table S1). Additionally, comparison of estimated TPs of fish species found in our streams extracted from FishBase indicate creek chub occupy the highest TP (Table S2).
From each stream, up to 20 creek chub were targeted and kept for analysis of stomach contents and stable isotopes. If less than 10 creek chub were caught during electrofishing, minnow traps baited with dog treats were placed at three points along the sampling reach overnight to increase sample size. Creek chub were euthanized using an overdose of buffered tricaine methanesulfonate and a dorsal muscle sample was taken for stable isotopes. Baseline samples, which included benthic algae as the aquatic baseline and leaf litter detritus as the terrestrial baseline, were collected from each stream at the time of fish sampling. Samples were placed on ice and then frozen at −20 °C. In lab, frozen fish muscle tissue plugs and baseline algae and detritus (terrestrial leaf litter) samples were thawed and a minimum of 0.1 g wet weight was retained from each sample (see Table S3 for sample sizes). Samples were then dried at 60 °C for 48–72 h and ground. Ground samples were then sent to the Jan Veizer lab at the University of Ottawa for quantification of stable hydrogen isotope ratios (δ2H) and to Great Lakes Institute for Environmental Research at the University of Windsor for quantification of stable nitrogen isotope ratios (δ15N) analysis.
Where possible, stomach contents were visually determined and recorded in the field. Stomach contents were used to categorize individuals into trophic groups (i.e., predators, omnivores, primary consumers) to complement isotope analysis and compare proportion of trophic groups present seasonally in each stream. Predators were characterized as individuals with only insects in their stomachs. Omnivores were characterized as any fish that had both plant (algae or detritus) and insect parts and/or worms in their stomach. It is noted that only one individual in all sites/seasons had presence of fish in their diet; this individual also had detritus and insects and was thus classified as an omnivore. Primary consumers were characterized by only algae or detritus present in their stomach, where algal primary consumers and detrital primary consumers were differentiated. Parasites were not considered in diet characterizations. The proportion of creek chub in each trophic group at each site and season was calculated as # of individuals in given trophic group/total # of individuals*100. Lastly, we calculated the mean proportion of omnivores and predators at each site across all seasons. ANOVA with Tukey honestly significant difference (HSD) post hoc analysis was conducted to test for significant differences in proportion of omnivores and predators between the three streams.
Fish sampling was conducted under a permit issued by the Ontario Ministry of Natural Resources and Forestry (permit #1086855) and an Animal User Protocol issued by the University of Guelph (AUP #3682). All statistical analysis was conducted in R (v.4.3.2).
Stable isotope analysis
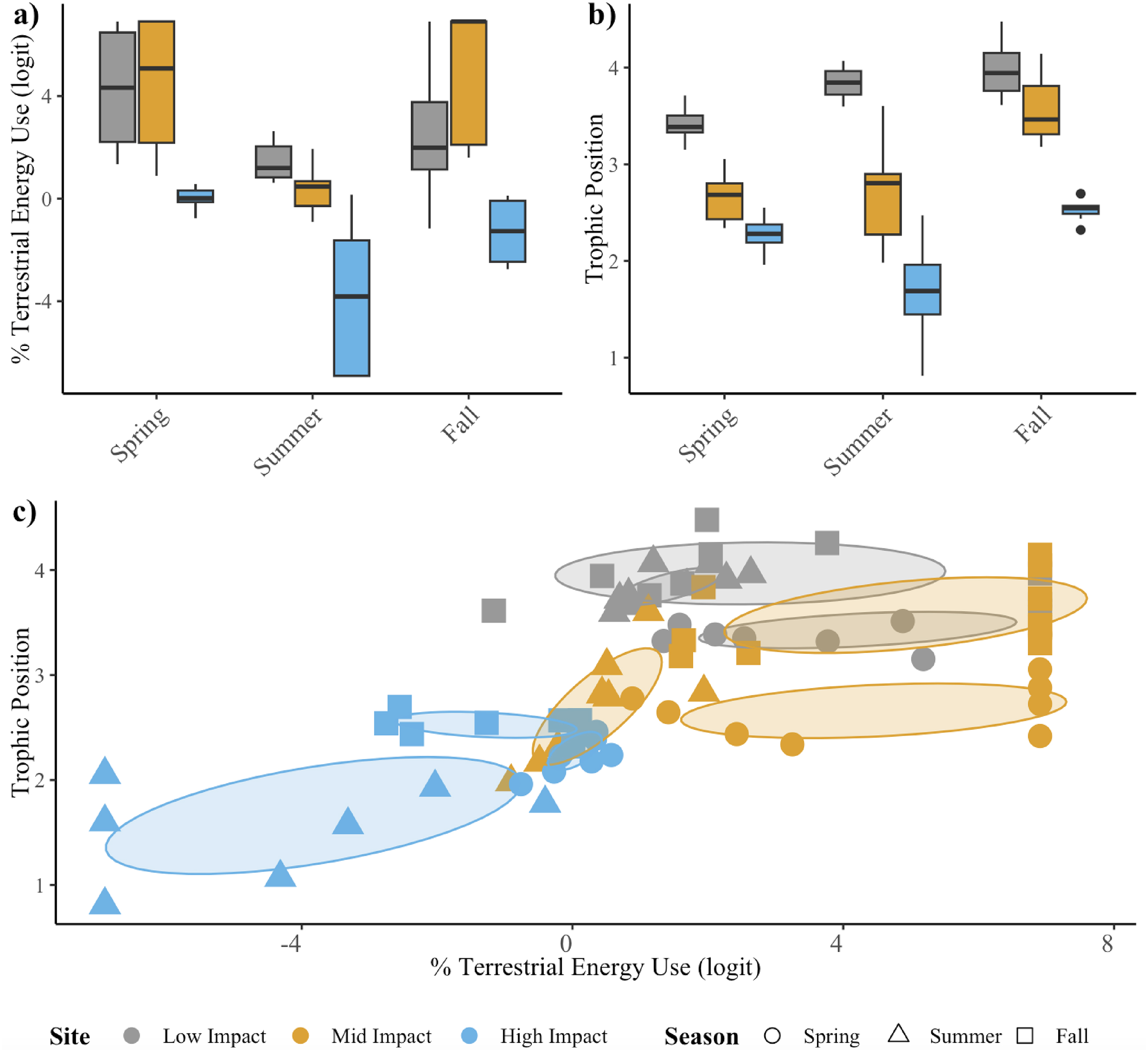
To understand how agricultural land-use change altered seasonal trophic structure of stream food webs, we analyzed seasonal patterns in three components of creek chub trophic structure: % TEU, TP, and trophic niche.
We calculated % TEU of each creek chub using δ
2H to determine how fish alter their foraging between terrestrial and aquatic energy pathways across seasons. As δ
2H can differentiate between aquatic and terrestrial food sources (
Doucett et al. 2007) they can be used to calculate the % TEU of generalist consumers. Prior to calculating % TEU, we adjusted the creek chub deuterium values to account for the contribution of environmental water to aquatic organisms’ δ
2H values (
Doucett et al. 2007;
Solomon et al. 2009;
Wilkinson et al. 2015;
Page et al. 2017). We used the following equations to adjust the raw creek chub δ
2H (
Page et al. 2017):
and
where
ω1 and
ω2 are the proposed proportion of environmental water in aquatic primary producers and secondary consumers, respectively, with
ω1 estimated to be 0.17 (
Solomon et al. 2009). As in
Champagne et al. (2022), we used the average δ
2H water value (−62.7‰) from two streams located within the Lake Erie watershed (
Gibson et al. 2020,
2021), as we did not collect water for isotopes at each site.
We then used the adjusted δ
2H values of each creek chub and the mean δ
2H of the algae and detrital baselines to calculate the % TEU of each creek chub using the following equation (
Vander Zanden and Vadeboncoeur 2002):
where
δ2Hcc is the adjusted value of the creek chub,
δ2Haq is the mean value of the aquatic baseline (i.e., algae), and
δ2Hterr is the mean value of the terrestrial baseline (i.e., detritus). Estimates ≤ 0 or ≥ 1 were set to 0.001 and 0.999, respectively, to maintain biological feasibility.
We calculated the TP of creek chub using δ
15N values. A two-source mixing model was used to determine the TP of creek chub using the following equation (
Post 2002):
where 1 is the TP of the baselines (algae and detritus),
δ15Hcc is the value of the creek chub,
δ15Haq is the mean value of the aquatic baseline (i.e., algae),
δ15Hterr is the mean value of the terrestrial baseline (i.e., detritus),
α is the % TEU of the individual creek chub, and 3.4‰ is the trophic enrichment factor.
To test for significant differences in % TEU and TP across season and site, we conducted two-way ANOVAs with a Tukey post hoc test in R. Histograms were used to graphically determine normality of data, Levene’s tests were used to test for homogeneity of variance, and outliers were determined as values ± 1.5 times the interquartile range. Subsequently, % TEU was logit transformed to improve normality and outliers were removed prior to ANOVAs.
To examine how agricultural land-use change influenced seasonal trophic niche space, we quantified the seasonal overlap of two-dimensional isotopic niche space based on TP and % TEU estimates. TP and % TEU (logit transformed) were used rather than raw isotope values as they are baseline corrected and thus provide more ecologically relevant information on food web structure (
Hette-Tronquart 2019). Standard ellipse area (SEA; ellipse containing 40% of data) and probability of niche overlap was calculated using the R package nicheRover (
Swanson et al. 2015). The package nicheRover uses a Bayesian approach to develop a posterior distribution of mean and variance for each group (i.e., each site by season), and we used 10 000 draws to develop estimates of SEA (
Swanson et al. 2015). We then calculated the mean and probability of overlap of 95% niche area for 10 000 Monte Carlo draws from the posterior distribution (
Swanson et al. 2015).
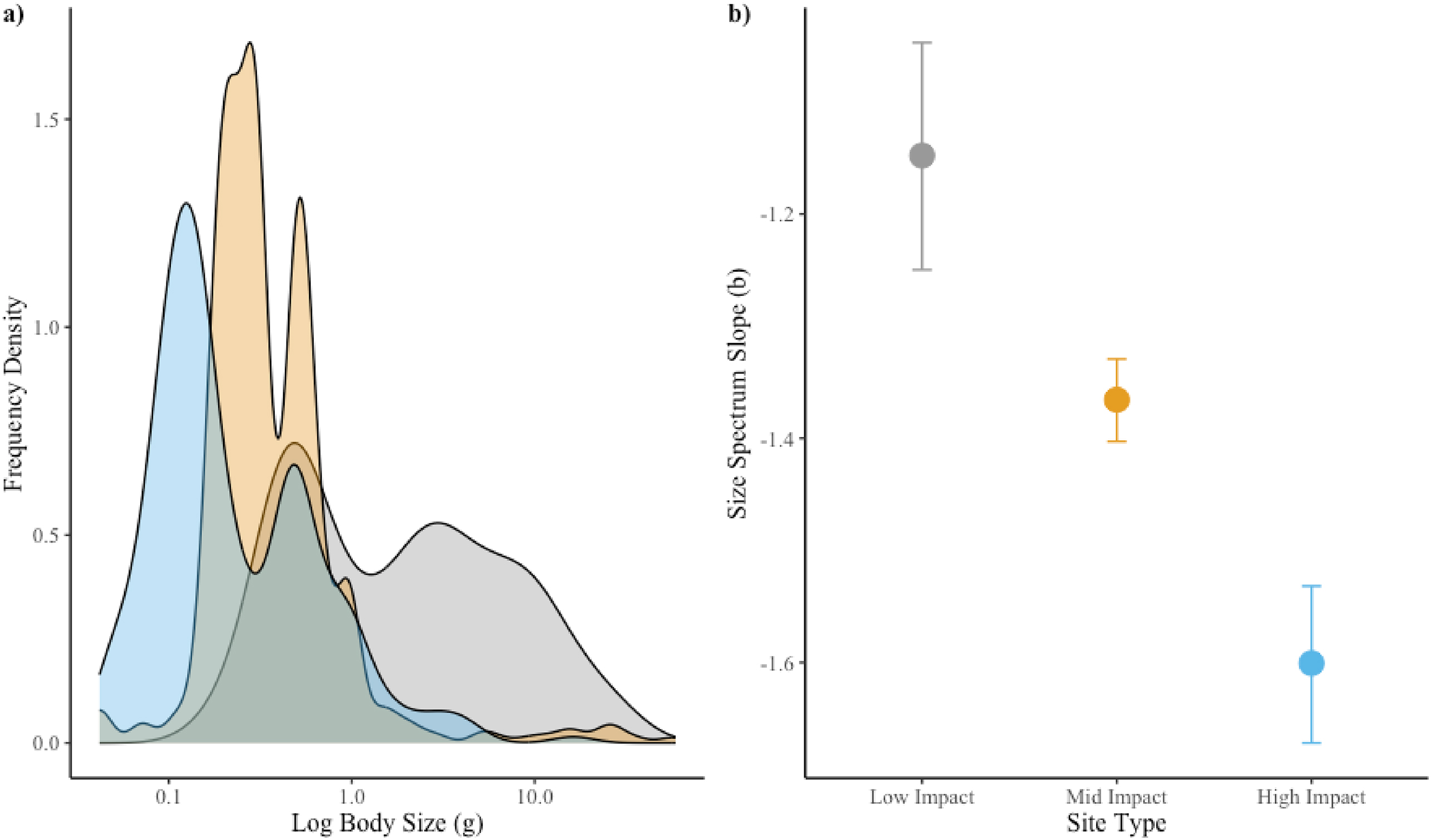
Fish community biomass structure
Lastly, to explore if agriculturally driven changes in seasonal food web structure altered fish community biomass structure we calculated seasonal and collective annual frequency density distributions and size spectrum slope (
b) of the fish community at each stream. Changes in fish community size structure are increasingly used as bioindicators of ecosystem health in freshwater ecosystems (
Marin et al. 2023).
For each stream, we calculated the individual size distribution (ISD) of all fish caught during electrofishing within each season (seasonal ISD) and across all seasons (collective annual ISD) using maximum likelihood size spectrum methods (
Edwards et al. 2017). We chose to use maximum likelihood methods as these have been shown to provide more accurate estimates of size spectrum slope than traditional binning-based regression techniques (
White et al. 2008;
Edwards et al. 2017). In this approach, the biomass of individual fish within each of our stream sites were fit to a bounded power law distribution with the probability density function:
where
x is the biomass,
b is the exponent of the power law relationship (size spectrum slope), and
xmin and
xmax are the minimum and maximum attainable body sizes, respectively (
Edwards et al. 2017). In our study,
xmin and
xmax were the smallest and largest individuals captured in each sample. The spring fish community at the high-impact site consisted of only six individual fish caught at two different sizes (two individuals at 0.5 g, and four individuals at 1.0 g). As it was not possible to generate an accurate slope estimate between two points, we added a single weight of 0.75 g to maintain the same qualitative pattern and allow for generation of a slope estimate. The size spectrum slope
b was estimated by maximum likelihood estimation using code modified from the
sizeSpectra package in R (
Edwards et al. 2017).
Discussion
Here, we hypothesized that agricultural land-use change would reduce terrestrial resource inputs and increase aquatic productivity, altering seasonal resource asynchrony and rewiring stream food webs towards highly productive, aquatic energy channel dominated food webs. The reduction in forest coverage and increase in agricultural land use at local and regional scales surrounding our stream sites suggest they ought to exhibit seasonal differences in relative terrestrial and aquatic resource availability. Given this, our results suggest these changes fundamentally alter the seasonal trophic structure of stream food webs through reductions in % TEU and TP that shift trophic niches and increase omnivory. Additionally, we found shifts in fish community biomass structure towards higher abundances of smaller fish under greater agriculture land-use change. Collectively, these results suggest agricultural land-use change drives dominance of the aquatic energy channel across seasons and shifts streams towards high productivity systems with faster turnover and potentially strong omnivory.
Importantly, the seasonal dynamics in trophic structure at the mid-impact site were more closely related to the low-impact site and exhibited the highest degree of seasonal switching across aquatic–terrestrial energy channels. These results highlight the potential for mitigation strategies that retain riparian structure and reduce nutrient loading to effectively retain seasonal dynamics that theory predicts contributes to the stability of stream food webs. However, the steeper size spectrum slope seen at the mid-impact site relative to the low-impact site indicate mitigation may be more nuanced, as theory has shown that communities with higher average growth rates (i.e., smaller, faster growing individuals) tend to have increased interaction strengths driving more unstable dynamics (
Anderson et al. 2008;
Rip and McCann 2011). Overall, our results suggest that agricultural land-use change can rewire the seasonal structure of headwater stream food webs in ways that may threaten their stability.
The land-use surrounding our three stream sites show a strong gradient in agricultural land use at local, riparian buffer, and watershed scales. Previous evidence from the literature has shown that removal of forested riparian buffers significantly reduced seasonal terrestrial subsidies (leaf litter and insect in-fall) into streams (
Kawaguchi and Nakano 2001;
Allan et al. 2003;
Little and Altermatt 2018;
Grunblatt et al. 2019). Additionally, there is extensive evidence that increasing agricultural land-use increases nutrient loading and aquatic productivity (
Smith et al. 1999;
Allan 2004;
Griffiths et al. 2013). In our sites, summer nutrient concentrations and % algae cover in our streams indicate the high-impact site had the highest aquatic productivity. However, the low-impact site had higher nutrient concentrations in the summer than the mid-impact site, suggesting surrounding agricultural land use may not always directly correlate to seasonal aquatic production. Improved seasonal measurement of aquatic and terrestrial resource availability and algal biomass is needed in future work. In general, the differences in land-use surrounding each stream suggest seasonal changes in resource availability, with reduced terrestrial subsidies and increased aquatic resource availability along our agricultural land-use gradient.
The seasonal changes in trophic structure observed here build on existing literature that has demonstrated similar changes (i.e., reduced %TEU and TP) in the mean response of stream foods webs across a larger gradient in agricultural land-use change (
Table 3;
Champagne et al. 2022;
Effert-Fanta et al. 2022). A recent study by
Champagne et al. (2022) investigated mean changes in food web structure (% TEU and TP) during a single season across 20 streams spanning a gradient in agricultural land-use intensity in the same geographic location as this study. Their study also included alternative land use services (ALUS) farms and importantly found that local riparian buffers played a key role in retaining food web structure even in the face of high agricultural land use in the surrounding watershed. Here, we see that these changes are consistent across season, where % TEU and TP are reduced with increasing agricultural land-use change. As in
Champagne et al. (2022) the changes in TP indicate increasing omnivory. We also see evidence of higher degrees of seasonal variability in the low- and mid-impact stream (i.e., higher degrees of switching across aquatic–terrestrial energy channels and/or reduced overlap in trophic niches across season). While research on mean stream food web responses to agricultural land-use change is growing (
Price et al. 2019;
Champagne et al. 2022;
Effert-Fanta et al. 2022,
2023), minimal work has examined agricultural impacts on seasonal stream food web dynamics. In this paper we provide important early evidence on how agriculture may be fundamentally rewiring temporal dynamics of these important ecosystems. Additionally, as seen in the mean response results, mitigation strategies that reduce nutrient loading and retain riparian buffers (such as those employed by programs such as ALUS) may be effective strategies to maintaining seasonal food web structure in streams.
Theory suggests the agriculturally driven temporal rewiring of stream trophic structure described above in conjunction with our observed shifts in fish biomass structure may threaten the stability of stream food webs. First, the increased reliance on the aquatic energy channel and reduction in seasonal switching across aquatic–terrestrial energy channels indicates a loss of seasonal asynchronous food web dynamics, that previously have been found to be an important stabilizing mechanism in stream food webs (
Nakano and Murakami 2001;
Takimoto et al. 2002). Second, the increase in omnivory suggests streams heavily impacted by agricultural land-use change may be exhibiting strong omnivorous interactions. While weak omnivory can be stabilizing to systems, theory has shown that strong omnivory tends to be destabilizing (
McCann and Hastings 1997;
Gellner and McCann 2012;
Gutgesell et al. 2022). Lastly, the shift in fish community body size distributions are consistent with existing research that has shown declines in species body size in response to agricultural land-use change (
Casatti et al. 2012;
Budnick et al. 2019) and climate change (
Sheridan and Bickford 2011). Allometric patterns in growth rate (
r) have shown historically and consistently that, as body size decreases, population maximal growth rates increase, necessarily driving increased interaction strengths (
Yodzis and Innes 1992;
Brown et al. 2004;
Rip and McCann 2011). Theory predicts that increases in interaction strength excite consumer–resource interactions into hostile dynamic overshoots that can drive the loss of either the consumer or the resource, therefore shifts in growth rate tend to lead to instability (
Yodzis and Innes 1992;
Rip and McCann 2011). Collectively, this suggests that agricultural land-use change is eroding stabilizing structures within food webs and potentially generating highly unstable temperate stream ecosystems.
While our results are consistent with current understanding of agricultural impacts on streams and theory, there are several limitations that limit generalizing our results to all agricultural systems. First, we only examine one stream at each agricultural impact level; by increasing the number of sites we can start to understand whether the patterns seen here are more broadly true. Second, we did not directly measure terrestrial inputs into streams or aquatic algal productivity. Instead, we used proportion of forest and agricultural land at local, riparian, and watershed scales surrounding our stream sites as a proxy of relative resource availability. While we did have some measurement of summer total nitrogen and total phosphorous levels, only the high-impact site showed evidence of increased N and P. Measuring N and P across all seasons and quantifying algal biomass would provide a more accurate estimation of aquatic productivity and resource availability. To explicitly test this mechanism future work should aim to seasonally quantify the relative biomass of terrestrial input and available aquatic biomass in streams with varying agricultural pressure.
In conclusion, we show that agriculture has the potential to seasonally rewire stream food webs by reducing terrestrial inputs and increasing within stream productivity. These changes can fundamentally alter trophic structure, driving highly productive, single-energy channel dominated food webs with smaller, faster growing fish communities. Theory shows how these changes in food web structure can drive instability by increasing interaction strengths and energy flow, suggesting agricultural impacts are fundamentally altering the stability of adjacent stream ecosystems. We also show seasonal variation in trophic structure and diet, demonstrating the importance of considering seasonality when examining impacts of agriculture and other types of global change on the structure and function of food webs. Finally, the retention of seasonal trophic structure in the mid-impact site highlights the important role of mitigation strategies and restoration programs that aim to maintain riparian structure and reduce nutrient loading for maintaining temporal stream food web structure in agricultural landscapes.